|
The new method uses the primer pairs labeled with different fluorophores, so that the multitarget DNAs in one mixed sample could be amplified in a single reaction set and then qualitatively and quantitatively analyzed on Infinite M1000 PRO. In this paper, we constructed and validated a novel high-throughput detection method based on combining emulsion PCR and fluorescence spectrophotometry (EPFS) for qualitative, quantitative and high-throughput detection of multiple biological genes. Emulsion PCR, alleviates nonspecific amplification in conventional multiplex PCR, increases the throughput and reduces the regent and sample consumption. In emulsion PCR, the different DNA targets are compartmentalized in millions of micro-reservoirs and parallelly amplified in a water-in-oil (w/o) emulsion. The multiplex emulsion PCR has been developed for high-throughput simultaneous amplification of several DNA targets, either used alone 18, 19 or combined with other methods 4, 9. Therefore, developing a novel qualitative, quantitative and high-throughput method for detecting multiple biological genes is of great importance. Poor linearity is another weakness of DNA microarray that limits its use in quantitative analysis 17. DNA microarray is a high-throughput approach used for analyzing complex nucleic samples that have limited feasible availability due to the complicated procedure and expensive consumption. Nonetheless, their throughput is low because of the limited number of channels in the real-time system. Real-time quantitative PCRs have been widely used to quantify target genes with high sensitivity, specificity and a wide dynamic range 13, 14, 15, 16. Indeed, several multiplex detection methods of target genes have been developed and employed in the fields of food/feed identifications 1, 2, 3, 4, medical diagnostics 5, 6, 7, and large scale sequencing 8, 9, 10.Īlthough conventional multiplex PCRs have always been used for amplification of nucleic acid samples, problems such as preferential amplification of shorter DNA templates, interference of multiple primer pairs and limited substrates limit their wide use in quantitative and high-throughput research 11, 12. Therefore, it is of great importance to develop an economical and more effective technique that would enable simultaneous qualitative and quantitative detection of these samples. 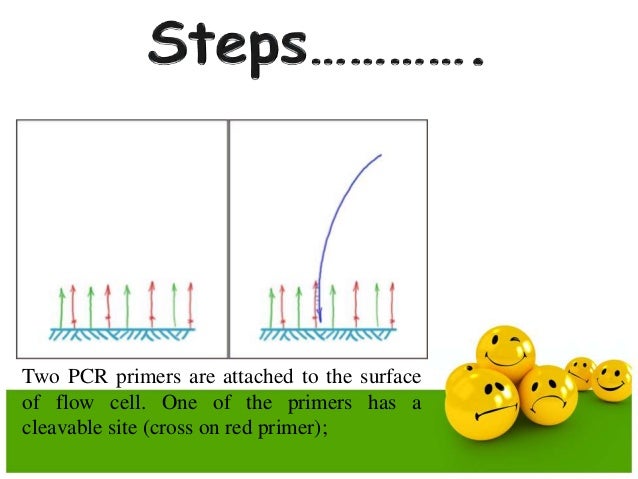
When dealing with complex nucleic acid samples, multiple reactions need to be performed separately, which is costly and time consuming. Traditional nucleic acid based technologies can usually detect only one target gene in one reaction. The increases in the number of biomolecular samples that need to be analyzed generate a great demand for a high-throughput detection method. To sum up, the new EPFS system is the first analytical technology of this kind that enables simultaneous qualitative, quantitative and high-throughput analysis of multiple genes. Consequently, the same qualitative, quantitative and high-throughput results were confirmed with the four GM maize. Moreover, the reproducibility assays were further performed using four foodborne pathogenic bacteria to further evaluate the applicability of the system. In addition, the quantitative results revealed that the absolute limit of detection was 10 3 copies, showing good repeatability. The qualitative results revealed high specificity and sensitivity of 0.5% (w/w). The sensitivity and specificity of the system was examined using four kinds of genetically modified (GM) maize. After product purification, different fluorescent-labeled DNA products were qualitatively analyzed by the fluorescent intensity determination. 
Through emulsion PCR, a target DNA was amplified in droplets that functioned as micro-reactors. In a single reaction set, each pair of primers was labeled with a specific fluorophore. We constructed and validated a novel emulsion PCR method combined with fluorescence spectrophotometry (EPFS) for simultaneous qualitative, quantitative and high-throughput detection of multiple DNA targets.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
